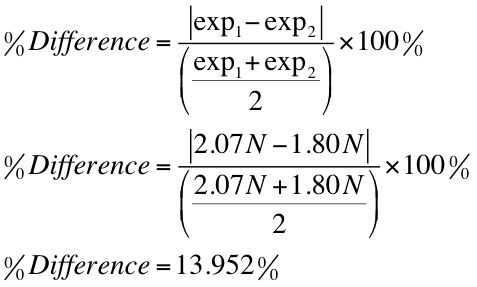
An object's mass also determines the strength of its gravitational attraction to other bodies. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. Mass/Volume Percent = 289.15 g C a ( N 3) 2 762.5 m L s o l u t i o n × 100. By using this chemists work out the chemical formula. How to use M r to calculate the percent mass of water in a compound?. In order to make the … How do you calculate percent by mass? For a solution, mass percent equals the mass of an element in one mole of the compound divided by the molar mass of the compound, multiplied by 100%. Mass is the quantity of matter in a physical body.It is also a measure of the body's inertia, the resistance to acceleration (change of velocity) when a net force is applied. Moreover, it describes the component during a particular mixture. 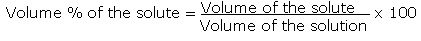
While the percentage of LBM is usually not computed, it on average ranges between 60-90% of total body weight. Now, the molecular weight calculator m ultiplies the atomic weight of each element by the number of atoms present in the compound. What is the percentage of carbon hydrogen and oxygen in glucose? 6. empirical formula = carbon 83.3/12 : hydrogen 16.7/1. Place your final answer in the FORMULA MASS COLUMN. The sum of all the mass percentages should add up to 100%. The mass fraction of a substance in a mixture is the ratio of the mass of the substance to the total mass of the mixture. Mass Volume percent calculator uses mass_volume_percent = (Mass of Solute / Volume of solution)*100 to calculate the Mass/ Volume percent, The Mass Volume percent formula is defined as the ratio of the mass of solute that is present in a solution, relative to the volume of the solution, as a whole. 
For a mixture, The fat mass percentage obtained with each of the skinfolds using the 3-compartment Siri's formula was compared to a reference value using the sum of the skinfolds. The numerator in the above formula gives the average atomic mass, which has a unit of the dalton or the unified atomic mass (u). Find the formula mass of the following compounds. Both low and high mass ranges can be selected. The equivalent weight of an element is the mass which combines with or displaces 1.008 gram of hydrogen or 8.0 grams of oxygen or 35.5 grams of chlorine. Percent composition in chemistry typically refers to the percent each element is of the compound's total mass. 
These values correspond to the atomic weight divided by the usual valence for oxygen as example that is 16.0 g / 2 = 8.0 g. Since the mass fraction is a ratio of mass to mass, it is a dimensionless quantity. Atomic mass of oxygen = 15.995 amu (99.76/100) + 16.999 amu (.04/100) + 17.999 amu (.2/100) It is also known as mass per cent or percentage by mass when expressing in percentage. The formula below for calculating body fat percentage was taken from research carried out in 1991 by Deurenberg which compared body mass index bmi and body composition. Let us look at the to two commonly used formula to find the mass percentage in a given question. A man with less than 75 percent LBM is said to be unhealthy, and for a woman, it is less than 68 percent. Steps for Finding The Empirical Formula Given Mass Percent. Examples: CuCl 2 => CuCl2 C 12 H 22 O 11 => C12H22O11 (C 6 H 5) 3 PCCO => C18H15PCCO Formula Find the ratio of the moles of each element. Mass Percent Formula - Mass percent is another method of expressing the composition of the solution mixture. Muscle mass is a part of your lean body mass. Denotation: Mass is represented by “M.” Weight is represented by “W." The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. Once you know your body-fat percentage, it’s easy to calculate your lean body mass (LBM). It shows the mass of solute present during a given mass of solution. But, since the abundance is in %, you must also divide each abundance value by 100.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
